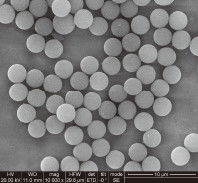
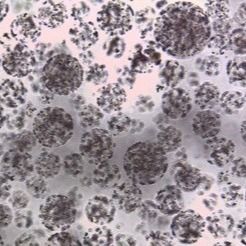
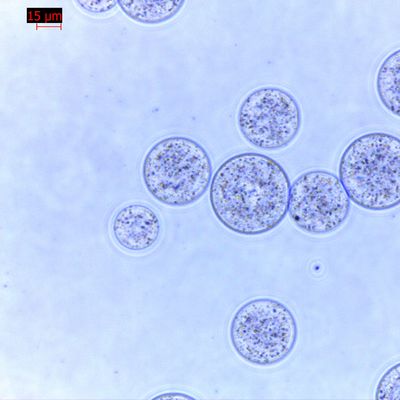
Achieving 95%+ Cell Purity in PBMC Isolation: Fast and Reproducible Magnetic Separation for U.S. Biotech Companies
Peripheral blood mononuclear cell (PBMC) isolation is a critical step in cell therapy research, immunology studies, CAR-T development, and clinical translational projects. For U.S. biotech companies, research institutions, and CDMOs, achieving high cell purity, strong viability, and reproducible results is essential for downstream applications such as T cell activation, immune profiling, and functional assays.
Traditional column-based methods often result in long processing time, inconsistent purity, and batch-to-batch variation. To address these challenges, BEAVER Biomedical Engineering Co., LTD. provides advanced superparamagnetic cell isolation magnetic beads designed for fast, column-free, and high-purity PBMC separation.
Why High-Purity PBMC Isolation Matters for U.S. Biotech
In highly regulated research and clinical environments, low cell purity or poor reproducibility can:
Compromise CAR-T and cell therapy development
Affect flow cytometry and immune profiling accuracy
Increase experimental costs due to sample loss
Create variability in GMP or preclinical workflows
Biotech decision-makers require solutions that offer:
≥95% cell purity
≥99% cell viability
Short processing time
Automation compatibility
Stable batch performance
Magnetic bead-based cell isolation technology provides a scalable and reproducible alternative.
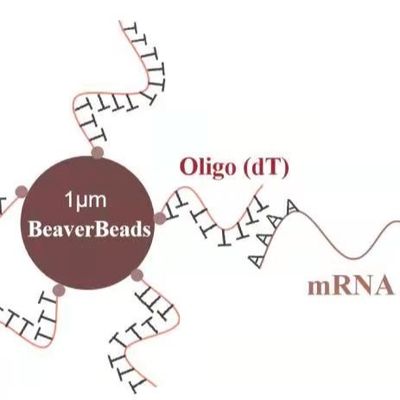
Magnetic Bead Cell Isolation Technology Overview
BEAVER’s superparamagnetic cell isolation magnetic beads enable both positive and negative selection methods for human and mouse immune cell separation.
Key Performance Specifications
| Parameter |
Specification |
| Magnetic core |
Fe₃O₄ superparamagnetic material |
| Particle size range |
300 nm – 5 μm (application dependent) |
| Magnetic response time |
<30 seconds |
| Cell purity |
≥95% (CD3/CD4/CD8 isolation) |
| Cell viability |
≥99% |
| Selection type |
Positive & Negative selection |
| Processing time |
As fast as 15 minutes |
| Storage temperature |
2–8°C |
| Shelf life |
Up to 24 months |
These specifications ensure consistent performance across research labs and production environments.
Faster PBMC Isolation Without Columns
Traditional Pain Points
Column-based separation methods often involve:
Flow rate control challenges
Sample clogging
Long centrifugation cycles
Operator-dependent variability
High equipment dependency
Magnetic Bead Solution
With magnetic bead separation:
No separation columns required
No need for pressure or flow-rate adjustments
Rapid magnetic response (<30s)
Simplified workflow
Scalable for high-throughput research
This makes it ideal for U.S. biotech companies, academic labs, and clinical research organizations focused on efficiency and reproducibility.
Achieving 95%+ Purity in T Cell Isolation
For applications such as:
CD3+ T cell isolation
CD4+ helper T cell purification
CD8+ cytotoxic T cell enrichment
CD34+ stem cell enrichment
CD3/CD28 T cell activation
BEAVER’s magnetic bead technology enables:
High-purity isolation exceeding 95%
Low non-specific adsorption
High cell activity after sorting
Reduced background signal
Reproducible batch performance
This ensures reliable downstream applications including cytokine assays, CAR-T engineering, and immune activation studies.
Designed for U.S. Biotech & Clinical Research Standards
U.S. research labs and biotech procurement teams prioritize:
Stable batch-to-batch consistency
Low non-specific binding
Short processing time
Compatibility with automated workflows
ISO-compliant manufacturing
BEAVER Biomedical Engineering Co., LTD. manufactures magnetic bead solutions under strict quality systems to support molecular diagnostics, immunoassay, and cell therapy research applications.
Benefits for Procurement & Engineering Teams
For purchasing managers and technical directors, magnetic bead-based PBMC isolation offers:
Reduced labor costs
Lower equipment investment
High throughput parallel processing
Reliable scalability
Long shelf life (24 months at 2–8°C)
By eliminating complex column systems, labs can reduce operational risk while increasing reproducibility.
Frequently Asked Questions (FAQ)
1. What purity level can be achieved in PBMC isolation?
Magnetic bead-based separation typically achieves ≥95% purity for CD3+, CD4+, and CD8+ T cells, depending on sample quality and protocol.
2. Is this technology suitable for cell therapy research?
Yes. The high viability (≥99%) and low non-specific adsorption make it suitable for CAR-T development, immune activation studies, and translational research.
3. Can the system support both positive and negative selection?
Yes. Both selection strategies are available to optimize experimental flexibility and preserve target cell integrity.
4. How long does the isolation process take?
PBMC isolation can be completed in as fast as 15 minutes, significantly reducing workflow time compared to traditional methods.
5. Are the magnetic beads compatible with automation systems?
Yes. The superparamagnetic properties and rapid magnetic response make them suitable for automated and high-throughput laboratory platforms.
Conclusion
For U.S. biotech companies seeking high-purity PBMC isolation, fast magnetic separation, and reproducible performance, advanced magnetic bead technology offers a reliable solution.
With optimized particle size, rapid magnetic response, low background binding, and stable long-term storage, BEAVER Biomedical Engineering Co., LTD. delivers scalable and efficient magnetic bead solutions tailored for modern cell therapy and immunology research.
If your lab requires consistent ≥95% cell purity with simplified workflows, magnetic bead-based PBMC isolation may be the upgrade your research platform needs.

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!